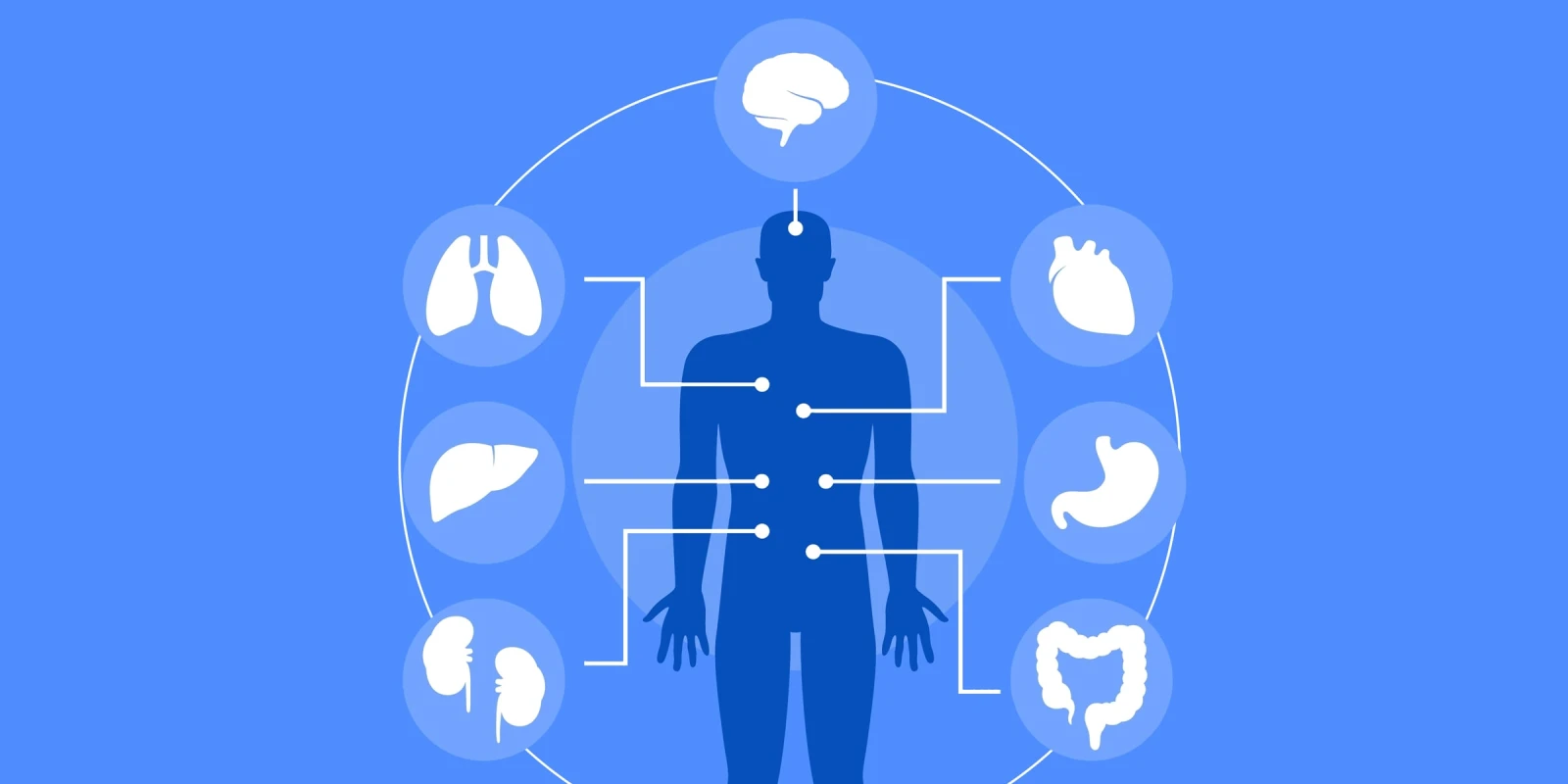
There is a moment familiar to every neurohospitalist — the patient who arrives for a stroke or seizure but whose most pressing problem in the next 48 hours turns out to be a crashing ejection fraction, a decreasing kidney function, or dysfunctional respiration, ventilation, and perfusion as medical complications pile up. The brain’s ability to receive the nutrients and oxygen it needs and the ability to get rid of toxic waste are pivotal for optimal function. That is why we need to be familiar with the management of these acute complications.
This concept sat at the heart of a compelling need to understand internal medicine conditions that affect our patient management as neurohospitalists. At this year’s American Academy of Neurology Annual Meeting in Chicago, I attended a three-part deep dive into internal medicine for neurohospitalists, covering respiratory failure in neuromuscular disease, cardiac complications of acute neurological illness, and nephrology considerations in critical care consultations.
Recognizing Respiratory Complications
Dr. Marin Darsie opened with a practical blueprint on recognizing impending respiratory failure and respiratory distress in neuromuscular disease. The takeaway was clear: re-examine the patient frequently to detect respiratory distress early, identify the mechanism of injury, use it to proactively work up the patient, and don't wait until blood gas parameters worsen before applying the appropriate intervention. By the time a patient with myasthenia gravis or Guillain-Barré is hypoxic on the monitor, the window for avoiding intubation may be closed. The "20–30–40 rule" — a negative inspiratory force worse than -20 cm H₂O, a vital capacity below 30% predicted, and a forced vital capacity (FVC) under 40% in conjunction with worsening single breath count (<15) or neck flexion/extension weakness — can be good indicators of impending respiratory failure that require intubation.
What resonated most for me was the emphasis on early BiPAP use. Evidence supports its use in myasthenic crises as a strategy that can meaningfully reduce the need for intubation. There was a practical reminder to titrate settings to the patient's work of breathing and to have goals-of-care conversations early. I personally have recommended BiPAP for hospital patients with myasthenic crises who are in respiratory distress, especially if the single breath count is between 16–24 and there is some mild neck flexion weakness. I watch the venous blood gas, FVC, and non-invasive ventilation trends carefully and if they do not progress, we can avoid intubation and complications. I tend to continue pyridostigmine and start IVIG right away except when it's a severe case (usually cannot avoid intubation here), then plasma exchange becomes the first choice for me.
The Heart's Response to Neurological Catastrophe
Dr. Stefanie Cappucci tackled cardiac complications with the kind of case-based energy that makes complex physiology stick. Stress-induced (Takotsubo) cardiomyopathy was a recurring theme — a condition whose diverse presentations (apical, midventricular, basal, focal variants) and strong association with subarachnoid hemorrhage, intracerebral hemorrhage, TBI, and status epilepticus make it a neurohospitalist diagnosis as much as a cardiology one.
The nuanced discussion of atrial fibrillation management in the stroke patient was particularly relevant. The temptation to reach for amiodarone in rapid ventricular response is understandable, but the data on its stroke risk — up to a twofold increase, especially without anticoagulation — argues for a more deliberate pharmacological approach. Another study showed that for new onset atrial fibrillation, beta blockers appear to be just as effective as amiodarone to achieve cardiovascular rate control, but digoxin and calcium channel blockers appear to be inferior. At times, cardiac electroconversion cannot be avoided for refractory cases or those with high risk of complications. On the other hand, magnesium, careful volume assessment, and addressing the underlying trigger (sepsis, pain, pulmonary embolism) can be considered before escalating pharmacological agents that may increase risk of stroke or bleed.
The Kidney Quietly Runs the Show
Dr. Erick Tarula’s nephrology lecture was the final piece of the session, where she reminded us that the kidney is an important variable in our management equations. For a field that leans heavily on osmolar therapy, renally-dosed antiepileptics, contrast-heavy imaging, and medications like vancomycin and acyclovir, the risk of acute kidney injury (pre-renal and renal mechanisms) is an important consideration for the practice of a neurologist. Also a consideration is the excessive use of contrast in our MRI, CT, and angiography studies to diagnose acute neurological conditions can be harmful for the kidneys especially in hypovolemic patients. Chronic kidney disease (CKD) is also a consideration, given increased risks of developing dementia, breakthrough seizures, and development of new onset medically refractory epilepsy associated with worsening CKD.
The discussion of continuous renal replacement therapy versus intermittent hemodialysis in patients was relevant given the implication in the pharmacokinetics of the medications we use for seizures and potential neurological harms when using erroneous dialysis strategies. Continuous renal replacement therapy’s gentler fluid shifts make it the preferred modality when increased intracranial pressure and reduced cerebral perfusion pressure is a concern. Monitoring osmolality closely and recognizing that dialysis will alter levels of renally cleared antiepileptics — levetiracetam (66% renal), gabapentin (100% renal), lacosamide (40% renal) — was a useful detail that keeps patients safe from having breakthrough seizures. As a practicing neurophysiologist with expertise in epilepsy, this is an important teaching point I will bring to the residents I supervise who take care of critically ill patients.
The Teamwork of Neuro
Neurohospitalists are a valuable community of practitioners with a distinct scope of practice, and that scope demands genuine fluency in internal medicine and critical care medicine. Many small community hospitals do not have dedicated neurointensivists or specialists to manage complex neurological patients. The academic neurohospitalist model may even have critically ill patients admitted under the general or stroke neurology attending teaching service. Despite having our counterparts help with ICU management, it is important that the neurohospitalist remains familiar with these medical complications and comorbidities to be better prepared to diagnose and treat.
The Neurohospitalist Society continues to improve as well, moving toward providing accreditation, structured core competencies, and curriculum development designed to train, grow, and train the neurohospitalist field. A compensation survey found that neurohospitalists are productive and often burned out (64% in one recent survey), and that we are still building the case for what this specialty is worth. The 2024 neurohospitalist compensation survey reveals a clear trade-off between workload and pay, though the data is limited by a small, geographically biased sample. Academic physicians work the fewest weeks (~20) for the lowest average salary (~$300,000). Non-academic physicians earn significantly more (~$367,000) but work about six weeks more per year. Locums, per diem, and self employed physicians, significantly underrepresented in the survey, command the highest earning potential, often making between $15,000 and $18,750 per week. We will have to see how pay continues to evolve. In the meantime, for those looking to stay sharp on their skills in the neurohospitalist field, the Neurohospitalist Society's expanding community of practice among program directors and its CME infrastructure can offer the education and support the field needs. These educational sessions will be needed more than ever as the field grows and expands. The brain, after all, is not an island.
Dr. Rocha Cabrero (double board-certified neurologist/clinical neurophysiologist, leader, activist, mentor, writer, son, brother, husband, father, he/him) is the CEO/owner of IRD Neuroanalysis Inc. Dr. Rocha is involved in different projects related to clinical medicine, writing, advocacy/policy, mentoring, and leadership across fields. His primary training focuses are epilepsy and neurointraoperative monitoring (NIOM). He also enjoys spending time with his infant son, traveling, dancing, EDM concerts, beach walks, and catching up with friends and family. Dr. Rocha Cabrero is a 2025–2026 Doximity Op-Med Fellow.
Image by vector next / Shutterstock